
a first-in-class treatment for open-angle glaucoma and ocular hypertension
OMLONTI® is a selective EP2 receptor agonist that reduces intraocular pressure by increasing both uveoscleral and trabecular outflow while preserving the integrity of periorbital tissue setting it apart from prostaglandins.
An EP2 agonist is different
OMLONTI targets both the trabecular meshwork and uveoscleral pathways.
Unlike FP agonists that are known to cause prostaglandin-associated periorbitopathy (PAP), OMLONTI has not shown to induce PAP-related side effects.
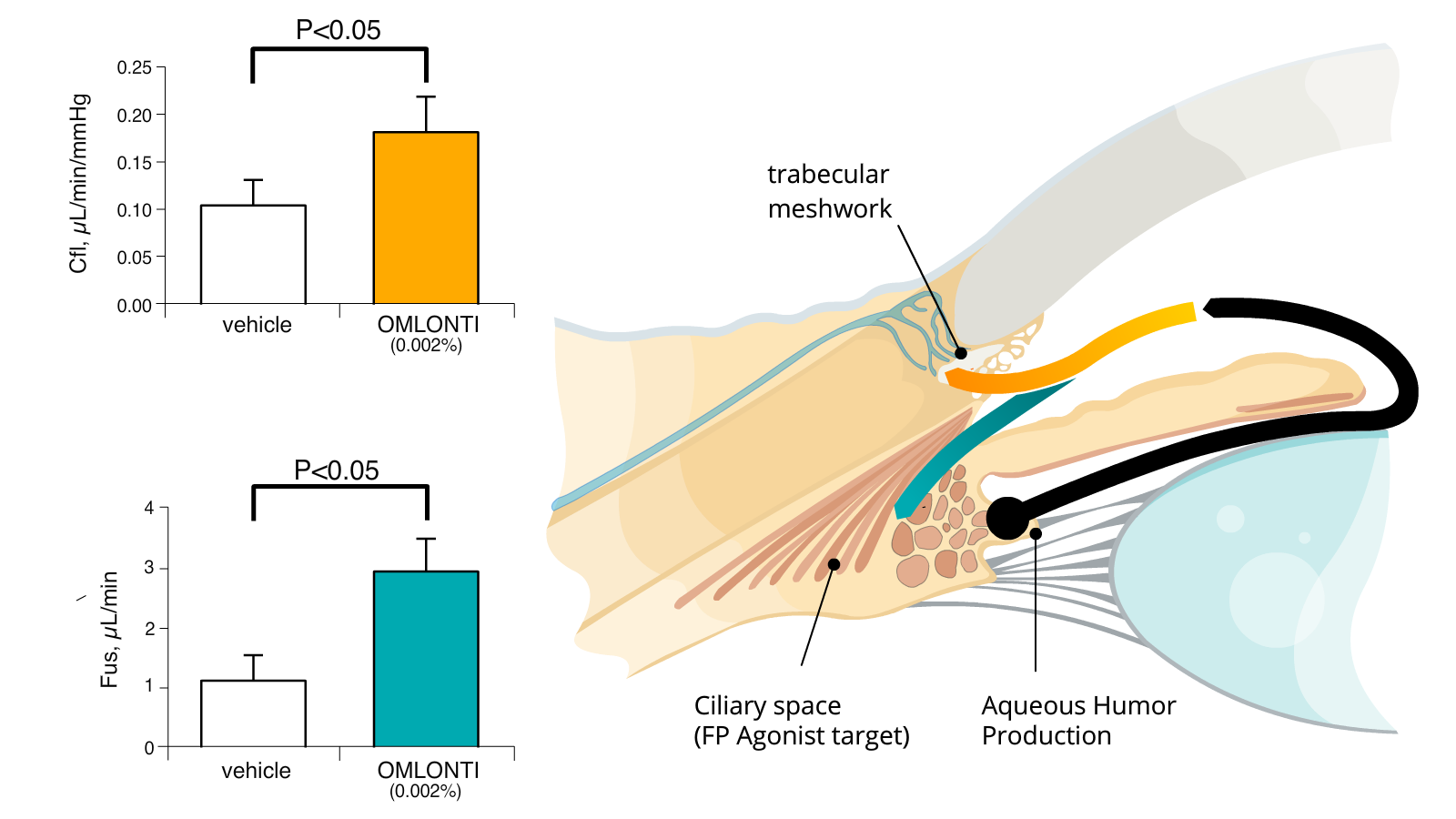
Omlonti has a dual mechanism of action
TRABECULAR OUTFLOW
Trabecular meshwork, or the Conventional Pathway accounts for 70%–80% of typically healthy eye outflows OMLONTI was shown to significantly affect these outflows.
UVEOSCLERAL OUTFLOW
Uveoscleral Outflow (intra ciliary space) or the Unconventional Pathway accounts for 20%–30% of typically healthy eye outflows OMLONTI was shown to significantly affect these pathways.
Omlonti Helps Patients that fail prostaglandins
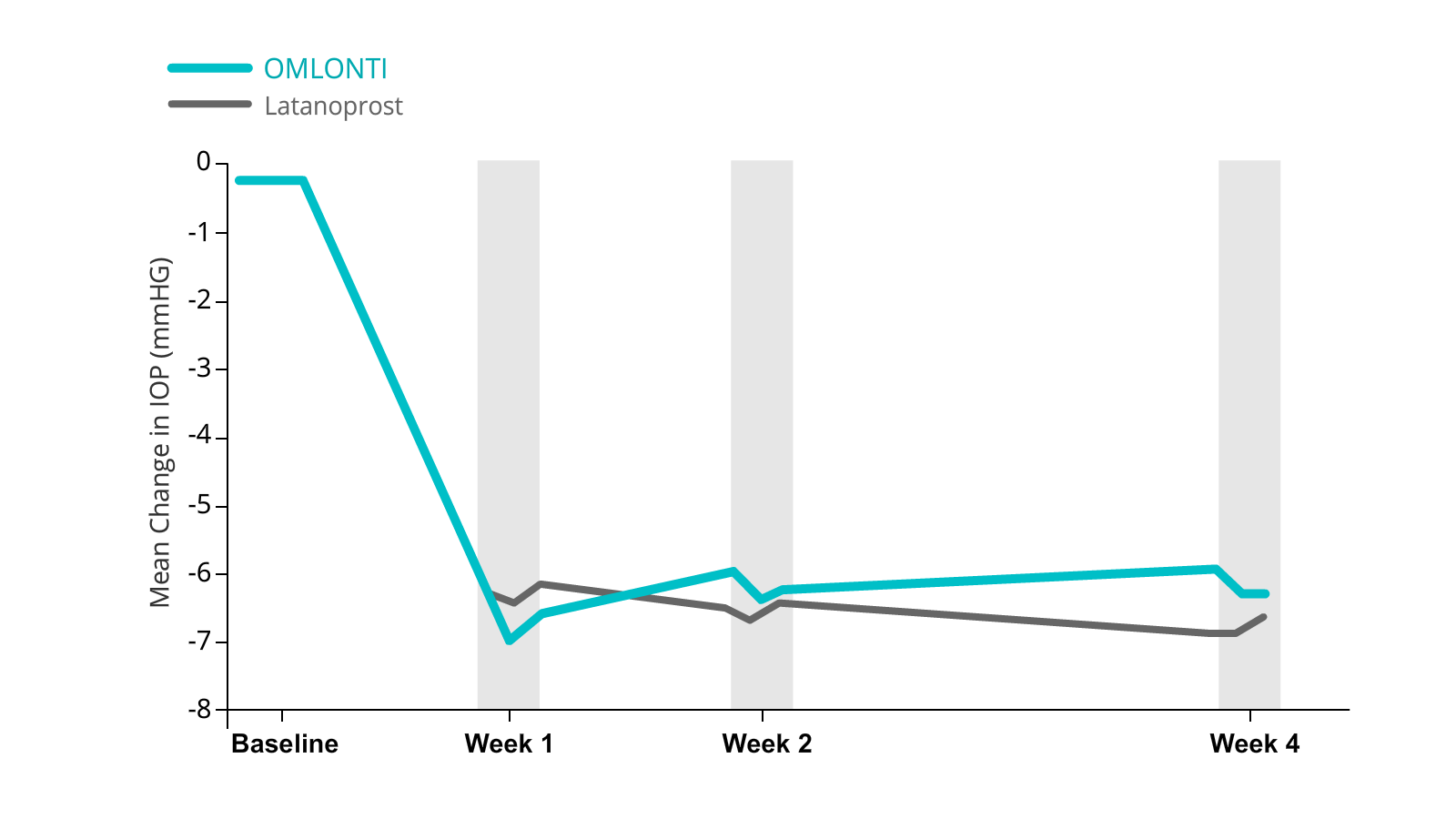
OMLONTI 0.002% was noninferior to latanoprost 0.005% in reducing IOP in patients with OHT or POAG and was well tolerated
Of the 190 patients randomized, 189 had at least 1 post-baseline IOP measurement. At baseline, patients who received OMDI or latanoprost had a mean ± SD diurnal IOP of 23.78 ± 1.73 mm Hg and 23.40 ± 1.51 mm Hg, respectively. At week 4, least-squares mean ± SE reduction in IOP from baseline with OMDI (−5.93 ± 0.23 mm Hg) was noninferior to that of latanoprost (−6.56 ± 0.22 mm Hg).
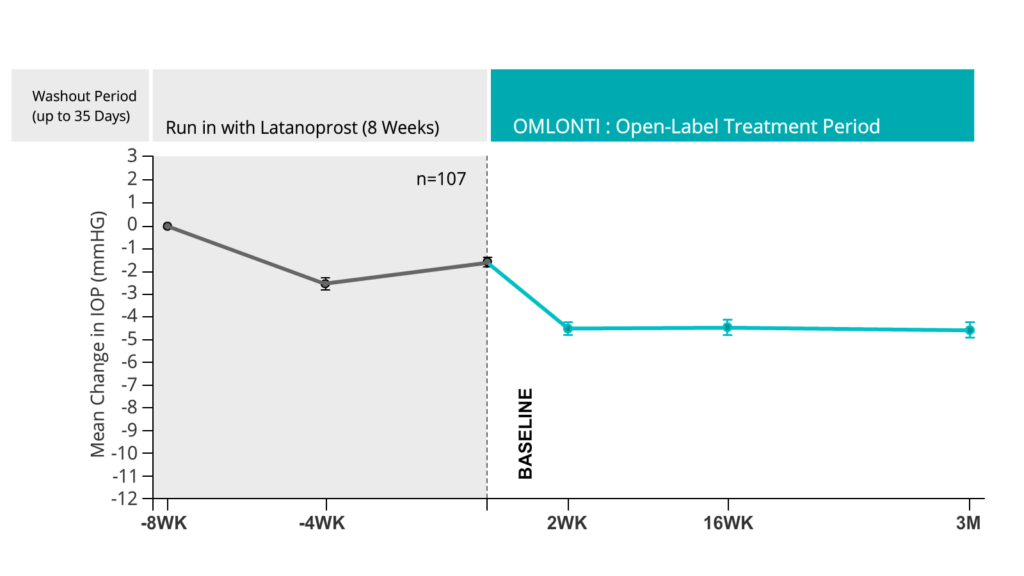
OMLONTI efficacy and safety in latanoprost low/nonresponders with POAG or OHT, suggesting OMLONTI is a treatment option in the patient population.
At baseline (visit 4), 75 (70.1%) patients had POAG, 32 (29.9%) had OHT, and 68 (63.6%) had prior use of prostaglandin/prostaglandin analogs (37.4% of whom used latanoprost). The mean (SD) baseline MD IOP was 23.34 mm Hg (2.12). The mean (SD) 3-month (visit 7) MD IOP change from baseline (following latanoprost run-in period and OMLONTI treatment period) was an additional decrease of 2.96 mm Hg (2.83) (P<0.0001). No significant safety issues were reported during OMDI treatment.
No Observed Prostaglandin-Associated Periorbitopathy
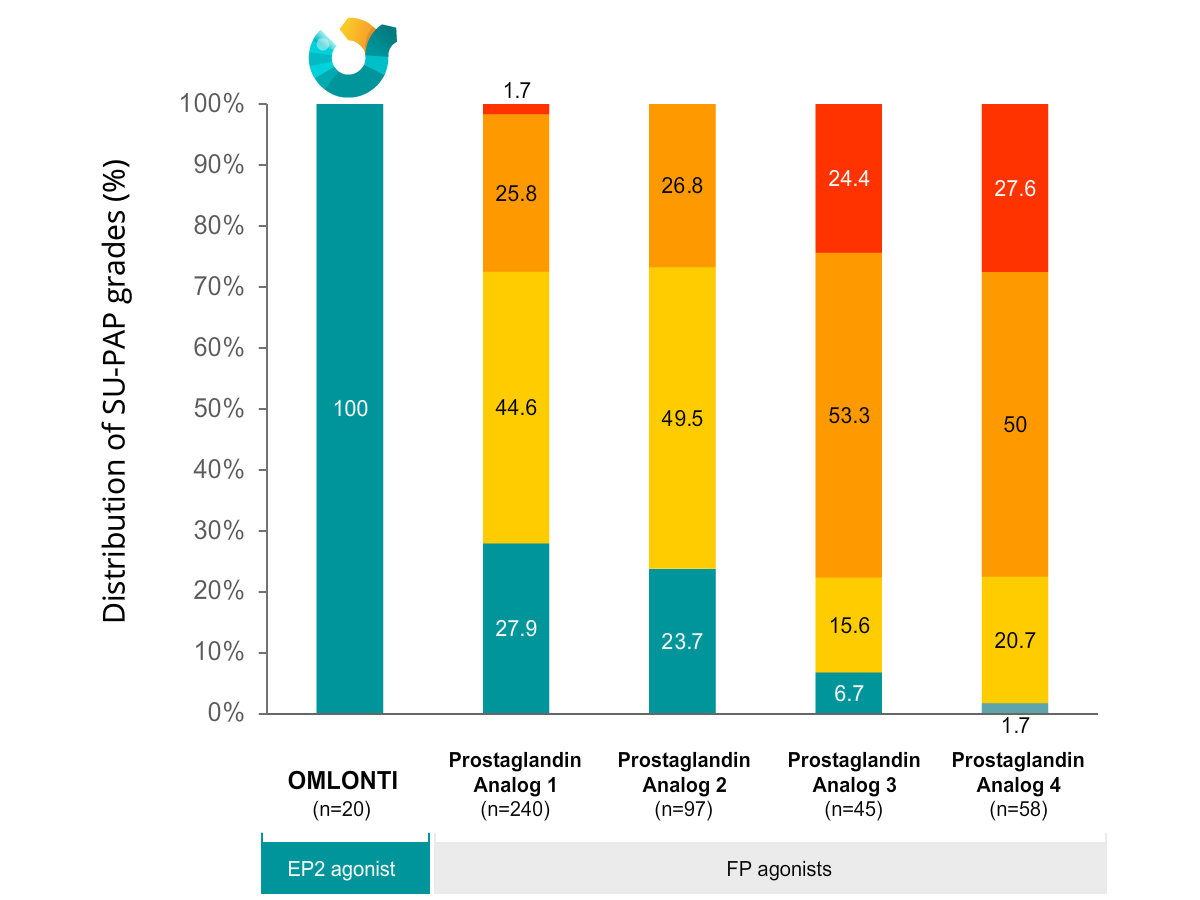
Grade 3
(Tonometric PAP, difficulty performing GAT and/or reduced reliability of GAT-measured IOP due to PAP-related DUES, hardening of eyelids, ptosis, or enophthalmos)
Grade 2
(Deep cosmetic PAP, cosmetic change(s) with at least one sign of PAP including DUES, blepharochalasis involution, periorbital fat loss, or enophthalmos)
Grade 1
(Deep cosmetic PAP, cosmetic change(s) with at least one sign of PAP including DUES, blepharochalasis involution, periorbital fat loss, or enophthalmos)
Grade 0
No PAP, no cosmetic change
References
Matsuo M, Matsuoka Y, Tanito M. Efficacy and Patient Tolerability of Omidenepag Isopropyl in the Treatment of Glaucoma and Ocular Hypertension. Clin Ophthalmol. 2022 Apr 26;16:1261-1279.
Tanito M, Ishida A, Ichioka S, et al. Proposal of a simple grading system integrating cosmetic and tonometric aspects of prostaglandin-associated periorbitopathy. Medicine. 2021;100(34):e26874.
Nakakura S, Terao E, Fujisawa Y, Tabuchi H, Kiuchi Y. Changes in Prostaglandin-associated Periorbital Syndrome After Switch from Conventional Prostaglandin F2α Treatment to Omidenepag Isopropyl in 11 Consecutive Patients. J Glaucoma. 2020 Apr;29(4):326-328.
